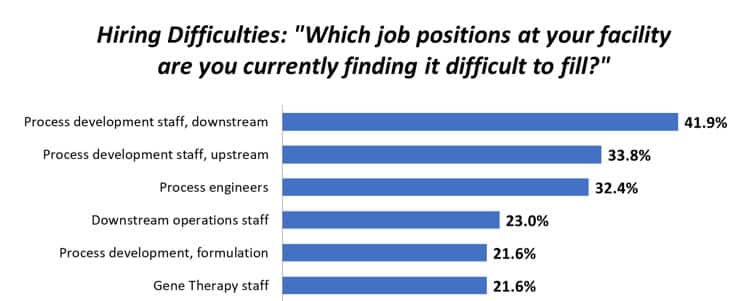
Research and development are vital for any biotech startup, however, raising venture capital (VC) can be a challenge for even the most experienced entrepreneurs. During November’s 2021 Biotech Start-Up Symposium, we were happy to have several VCs, industry leaders and experienced attorneys join us to provide key insights on what startup pioneers should know before pitching to a VC firm.
Tip 1: Know Thy Investor
Start by creating a potential roster of VCs that have a history of investing in the biotech space that you are part of. Pitching to a VC firm without sector experience in the biological arena is risky. Chances are they don’t know the ins and outs of getting a drug or device to market – let alone the science behind your idea. According to Dr. Anique ter Braake of M Ventures, the corporate strategic venture arm of
“These are long-term relationships – akin to a marriage.”
~Dr. Anique ter Braake, M Ventures
Tip 2: Put Yourself in the Shoes of the Investor
During the symposium, Matthew Meyer, Chief Business Advisor at the law firm Wilson Sonsini, said VC firms look at more than just your idea. He said, “Investors want to know why this opportunity? Why you? And why now?” Investors are more likely to trust their money with companies and individuals with deep domain expertise, a passion for solving problems and working in a big market.
Dr. ter Braake reminded attendees of the symposium that “as with many walks of life, you mostly will have only one chance to make a first impression.” So come up with a strong point plan and positioning strategy while also having a detailed plan for what your investor’s money will be used for if they choose to invest with you.
“Investors want to know why this opportunity? Why you? And why now?”
~Matthew Meyer, Wilson Sonsini
Tip 3: Don’t Oversell Your Idea
You love your idea and have spent countless hours making it a reality. But keep in mind that it’s easy to get caught up in the excitement and prospects of your own idea. Ms. ter Braake reminded attendees of the symposium that overselling your idea will set off alarm bells that you will not be able to un-ring.
She went on to say, “When VC firms hear statements like “We will see no side effects at all” or “There is no competition,” they are immediately skeptical. In most cases, VCs you meet have already done a quick analysis of the opportunity hence know more than what they may show at the meeting. Chances are that someone else has thought of something similar or another group of scientists are working to solve the problem in a different way. It’s important to be confident but stay factual. Investors want to know what is different about your idea, what problem it will solve, and why you are the best team to deliver that.
Tip 4: Communicate! Communicate! Communicate!
When looking outward for funding don’t forget to look inward at how your team is communicating with one another. Dr. Johnson reminded attendees that an effective intellectual property (IP) strategy means getting all team members on the same page. Ensure your entire team attends regularly scheduled meetings to discuss the latest developments and to make each department aware of what is going on elsewhere. Working in closed-off silos will lead to problems downstream. But regularly scheduled meetings between your R&D team, management, IP counsel and anyone else on your IP strategy team is key to VC success.
“An effective ongoing IP strategy should involve open communication.”
~Dr. Lee A. Johnson, Wilson Sonsini
Tip 5: Expect the Unexpected
You’ve done your research, scheduled a meeting with a potential investor and are about to pitch your idea. Suddenly, the electricity goes out in the conference room and the pitch deck you’ve spent so much time on can’t be shared on the big screen.
It sounds silly. But these things happen. Anyone who has attended a Zoom meeting recently knows that audio and visual issues are common. The best defense is to know your material cold. You and your team have to know your data like the back of your hand, so it can be shared no matter where you are or who you are with. Mr. Meyer said it best when he said “Remember that this is hard work. Try to embrace it.”
One Last Thing
Before finishing her session, Dr. ter Braake shared one last piece of advice for biotech pioneers: “Keep at it. Just because they said “no” the first time doesn’t mean they will the second or third time.”
“Keep at it. Just because they said “no” the first time
doesn’t mean they will the second or third time.”
~Dr. Anique ter Braake, M Ventures