| Single-source manufacture of biopharmaceuticals is becoming more and more rare. Increasingly, biomanufacturers or “sponsor” companies partner with contract manufacturing organizations (CMOs) to benefit from the CMO’s technology, equipment, and years of expertise. This collaboration helps the sponsor company mitigate risk, get their molecule to patients faster, and manage costs.
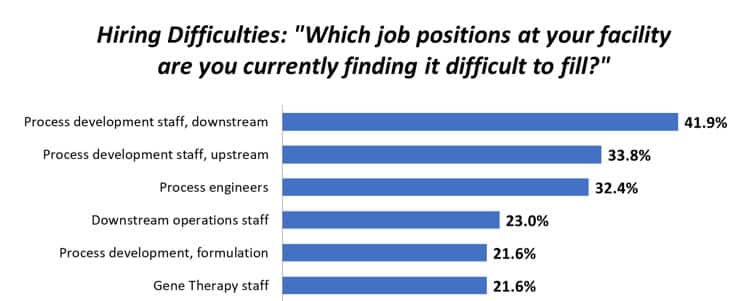
An obvious challenge to this collaboration is prompt and dependable sharing of data back and forth between sponsor and CMO. While many sponsor companies and CMOs have made advances in their digital transformations these advances remain within their own organizations and not between companies. What’s your problem? Generally, data are shared between sponsor and CMO via email. Gathering the data is a time-consuming, manual effort which leads to errors, rework, cost escalation, potential data integrity problems, and project delays. In an industry where real or near real-time data are essential to early issue detection and avoiding quality problems, late or slow data degrade in value. Lack of timely data sharing is also detrimental to compliance and hinders regulatory activities such as continued process verification (CPV). So, how do sponsors and CMOs realize the advantages of their partnership while avoiding the pitfalls of clunky data sharing? The BioPhorum’s DISCO In November 2022 the BioPhorum’s Digital Integration of Sponsor and Contract Organization (DISCO) workstream published Vision for Digital Maturity in the Integration Between Biomanufacturers and Partner Organizations. This document outlines the need for better data sharing between sponsor and CMO, puts forth the principles for digital integration, and lists benefits such as: · Speed – data access yielding faster issue resolution · Quality – reduce manual tasks with fewer errors · Value – optimize work processes · Agility – process improvement analytics This could help Fortunately, technology exists that enables better collaboration and communication between sponsor and CMO. There are available on the market software tools that automatically acquire and aggregate data across an entire bioprocessing workflow thus eliminating the error-prone, manual data gathering. Data can then be shared via a secure intranet or the cloud. Some software can also share real or near real-time visibility of in-progress and completed batches. Finally, these tools also can provide advanced analytics to help you make sense of all this data and support your decisions. Reference: BioPhorum DISCO Workstream, Vision for digital maturity in the integration between biomanufacturers and partner organizations, 30 November 2022 |