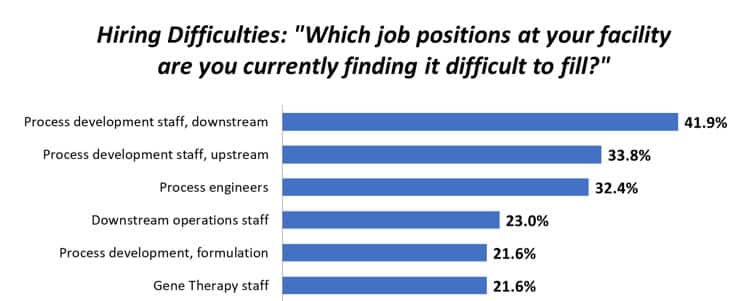
Depending upon your perspective, “emerging” biotech could mean any number of things, ranging all the way from the recent spinout of an academic lab to a publicly held company developing a therapeutic that has not yet reached the marketplace. My company, Respana Therapeutics (a finalist in
Thrilling you get, right? Cutting edge science, the potential for a big payout, etc. But anxiety-inducing? There are numerous reasons for this. Some of the key ones fall under the heading of gaps: technical gaps, gaps in funding, and gaps in expertise.
Technical gaps are familiar to anyone working in a biopharma firm, large or small. Examples include success at the bench that proves elusive at scale, manufacturing that’s projected to be cost-prohibitive, or failure of a clinical trial in Phase I, II or (ouch) III.
Potential gaps in funding are also familiar to most, regardless of company size. As of this writing, in early November, those working in well-established biopharma companies are probably in the middle of the budget review process. This annual vetting of development programs invariably necessitates tradeoffs.
But if you’ve never worked in venture capital or a startup you may not have experienced the “Valley of Death.” This refers to the point in a young company’s life cycle wherein the firm is ready to move further and faster than government grant funding, sweat equity and so-called “friends and family” money can take it buts lacks sufficient preclinical data to attract private investment.
Gaps in expertise can also be a challenge. This is in no way an attempt to undersell the work of academic labs! Respana Therapeutics and countless other firms contributing to global pipeline of novel therapeutics wouldn’t exist without the vision of so many working within academic and not-for-profit research institutions. A shout goes out here to Dr. Zissis Chroneos, Respana’s scientific co-founder, who has dedicated decades to the study of respiratory illness and whose insights into surfactant protein A receptor SPR-210 stimulated development of Respana’s therapeutic mAb. Invariably, however, the skills that lead to success in an early stage research environment will require complementary expertise in translational science and commercialization. (See a related post to this forum from Barri Blauvelt.)
Addressing these gaps is where programs like
Despite the challenges of working within this stage of the company life cycle, many of us who work in early stage life sciences firms wouldn’t want to do anything else. We do this work because of a passion for the science, the drive to improve healthcare, or both. If you’ve read this far, you probably feel the same way – so, let’s get back to work. And if you happen to be part of, or are contemplating starting, a life sciences startup in Pennsylvania, here are some additional, regionally based resources that Respana Therapeutics has found helpful: