We invite you to the 4th Annual Biotech Start-up Symposium, a free, two-day event to help Biotech Start-ups navigate their path to commercialization. This Symposium features industry experts who present and discuss various aspects of emerging technologies and the latest trends in their fields.
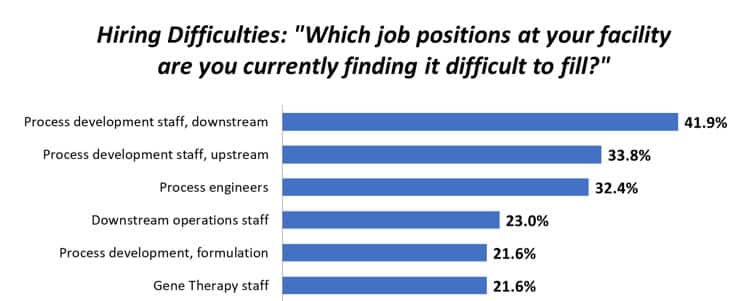
On November 14th, Day 1, you won’t want to miss the presentation from Natika Calhoun on Regulatory Considerations: Use of Ancillary Materials in Biologics.
Here is an overview of what is discussed:
What are the expectations for using qualified raw materials in developmental phases? There are many definitions, especially for novel modalities like Cell and Gene Therapies. Key definitions are evolving across agencies, and it’s necessary to understand regulations, guidelines, and international standards.