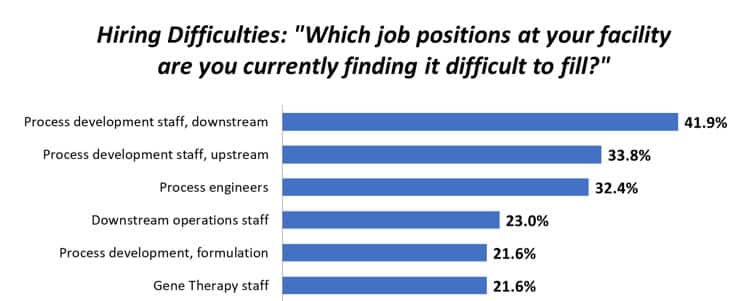
Click HERE to watch the On Demand Event.
Abstracts
Building a Successful mAb Platform using the CHOZN® Cell Lines and UCOE® Technology
Steven Richardson, Technology Consultant, Upstream Bioprocessing
Developing a mAb expression platform is a critical and time-consuming task. The CHOZN® Expression Platform includes a ready-to-use, industry-leading CHO cell line that creates high-producing clones without needing process optimization.
Join us for an introduction to the CHOZN® cell line and how to bring your project from DNA to GMP production. We will also share information on our UCOE® technology, which improves plasmid integration and mAb expression levels to drive higher titers.
ADCs and the Growing Atlas of Bioconjugates
Kaelyn Wilke, Associate Director, ADC & Bioconjugation Services
Deemed “magic bullets” by some in the biotherapeutics community, antibody-drug conjugates (ADCs) chemically link the potency of small-molecule chemotherapies with the selective targeting of monoclonal antibodies (mAbs). Recently, non-traditional ADC components across the small-to-large modality space are used to form novel conjugates and reach new and diverse indications.
In this presentation, we will explore this growing atlas of bioconjugates.
From Discovery to Market Readiness, a CTDMO as an Enabler
Jean François Michel, Head of Commercial Manufacturing, Life Science Services
Jean-Baptiste Noirot-Cosson, Head of Manufacturing Science and Technology, Life Science Services
In early development, biotechs are challenged to move fast while generating data on their lead molecule and its manufacturability. How do you make sure that process data isn’t lost in translation between clinical and commercial manufacturing?
Here we present how biologics contract testing development and manufacturing (CTDMO) can be an enabler for innovators to bring their molecule to market. We’ll discuss the importance of tech transfer for market readiness, the impact of harmonized single-use equipment on faster validation, and our commercial production facility’s capacity and capabilities.
Click HERE to watch the On Demand Event.