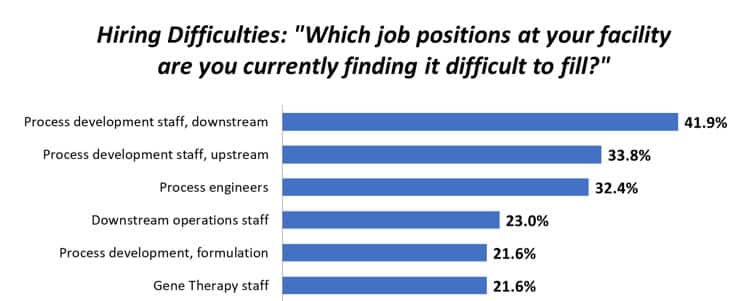
The tangential flow filtration (TFF) unit operation in viral vector manufacturing is a critical step on the path to commercialization. In this episode, Ratish Krishnan and Akshat Gupta discuss best practices – and common misconceptions – when establishing process conditions and utilizing different TFF device formats.
Below are some of the questions discussed during the podcast:
- How and why is tangential flow filtration (TFF) used in viral vector manufacturing?
- What are the main types of TFF formats and membranes used in viral vector purification?
- What are the key factors and best practices when it comes to designing a TFF step?
Learn how best practices can be applied on different TFF formats to overcome common concerns in viral vectors.
Click HERE to access the full content